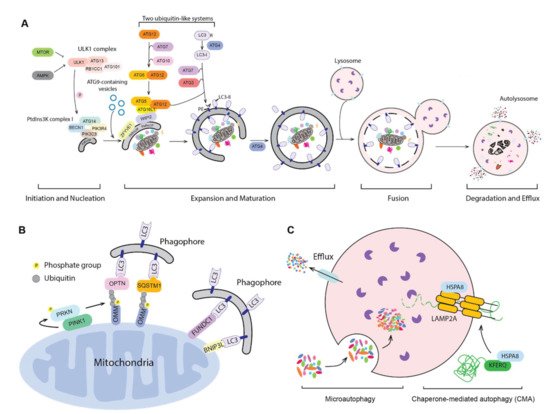
Many external stimuli may affect autophagy in cancer, such as hypoxia, acidification of tumor microenvironment, nutrient deprivation, drug therapies, or infections. In different stages of tumorigenesis, autophagy may have opposite and context-dependent roles acting as tumor suppressor or cancer promoter. However, the excessive stimulation of autophagy can be considered a self-eating mechanism favoring cell death and the aberrant regulation of autophagy is widely studied to improve therapeutic treatments for various diseases including cancer. Accumulating evidence also demonstrates that autophagy declines with age and that impaired autophagy favors individuals to age-related diseases, whereas interventions that stimulate autophagy can promote longevity. In this way, autophagy induces the degradation and recycling of cellular components to support energy metabolism in stressing conditions and cellular homeostasis. Furthermore, we summarize the molecular mechanisms and the cellular effects of GAPDH aggregates, which are correlated with mitochondrial malfunctions and can be considered a potential therapeutic target for various diseases, including cancer and neurodegenerative disorders.Īutophagy is a physiological degradative mechanism of the cells by which autophagic vesicles deliver unfolded proteins and damaged organelles to lysosomes for their elimination. Since autophagy is a degradative pathway associated with the regulation of cell death, we discuss recent evidence supporting GAPDH as a therapeutic target for autophagy regulation in cancer therapy. Here, we elucidate the correlation between autophagy and GAPDH in cancer, describing the molecular mechanisms involved and its impact in cancer development. In disease, the knowledge of the mechanisms regarding GAPDH-mediated cell death is becoming fundamental for the identification of novel therapies. Non-glycolytic functions of GAPDH include the regulation of cell death, autophagy, DNA repair and RNA export, and they are observed in physiological and pathological conditions as cancer and neurodegenerative disorders. The GAPDH functional diversity is mainly due to post-translational modifications in different amino acid residues or due to protein–protein interactions altering its localization from cytosol to nucleus, mitochondria or extracellular microenvironment. Several studies indicate that the cytosolic enzyme glyceraldehyde-3-phosphate dehydrogenase (GAPDH) has pleiotropic functions independent of its canonical role in glycolysis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
